Side Effects In Children
Side effects of Lantus in children are similar to those seen in adults.
However, in clinical studies of people with type 1 diabetes who took Lantus, children had more episodes of hypoglycemia than adults. In the 26-week study, 23% of children had at least one episode of severe low blood sugar. This was compared to 10.6% of adults in a 28-week study.
If youre a parent or caregiver of a child who takes Lantus, know the symptoms of hypoglycemia. This will help you notice when your child needs to consume foods or drinks to quickly raise their blood sugar. By aiding your child right away, you can help prevent a medical emergency.
Is It Better To Take Lantus In The Morning Or At Night
The best time to take Lantus depends on how your blood sugar levels change throughout the day.
In clinical studies, blood sugar levels were reduced to a similar degree whether people took Lantus in the morning or evening.
Your doctor will monitor how your blood sugar levels change throughout the day. Then theyll recommend the best time for you to take Lantus.
Kwikpens Tempo Pens And Needles
Basaglar comes in a prefilled, disposable KwikPen. The KwikPen comes in packages that contain five pens.
Youll inject your dose of Basaglar using the KwikPen or Tempo Pen and a needle. Needles arent included with the KwikPen or Tempo Pen. The manufacturer recommends using BD brand needles with your pen. Needles will be available for purchase at your local pharmacy.
The KwikPen and Tempo Pen are meant to be used more than once. The number of times you use your pen will depend on your dosage. You can use the KwikPen or Tempo Pen up to 28 days after you first open it. After 28 days, you should throw away the opened pen, even if it still contains medication.
Unopened Basaglar KwikPens and Tempo Pens should be stored in the refrigerator until youre ready to use them. Once youve opened a pen, it should be stored at room temperature, for up to 28 days. For more details on storing Basaglar KwikPens and Tempo Pens, please see the Basaglar expiration, storage, and disposal section above.
Needles are meant to only be used once. Youll need to use a new needle for each injection of insulin.
Always use a new needle each time you inject Basaglar. Never share needles with someone else. Using new needles helps prevent the spread of germs, which decreases your risk of infection.
Read Also: Glucagon Sequence
If Im Taking Lantus Can I Switch To Basaglar
Yes, youd be able to make the switch if your doctor recommends that you change treatment from Lantus to Basaglar.
Lantus and Basaglar contain the same type of insulin: insulin glargine. The Food and Drug Administration has determined that Basaglar is a follow-on insulin glargine to Lantus. This means that Basaglar is very similar to Lantus and that its just as safe and effective as Lantus.
Basaglar and Lantus are both available in the same strength: 100 units of insulin per mL of drug solution. If you switch from Lantus to Basaglar, your dosage will probably stay the same.
If youre interested in switching from Lantus to Basaglar, talk with your doctor to see if thats a good option for you.
How To Take Basaglar
You should take Basaglar according to your doctor or healthcare providers instructions.
Basaglar is taken as an injection under your skin . When you first get your Basaglar prescription, your healthcare provider or pharmacist will explain how youll inject the medication.
The manufacturer of Basaglar provides step-by-step instructions for both the Basaglar KwikPen and Basaglar Tempo Pen.
They also provide a video with instructions for use. Theres also an app called Beginning Basaglar, which is available on iPhone/iPad and Android devices. This app lets you download medication instructions and helpful tips for using Basaglar.
Note: Making changes to your insulin treatment plan could increase your risk for both hypoglycemia and hyperglycemia . These changes include taking your insulin differently than you usually do, starting a new insulin drug, or using a new insulin dosage. In addition, injecting your insulin into areas of skin that are thickened or pitted can also affect your blood sugar level. Be sure to talk with your doctor before making any changes to your insulin regimen.
Don’t Miss: Metformin Er 1000 Mg Side Effects
Consistent A1c Reduction And Demonstrated Safety In A Broad Range Of Adult Patients1
Select a patient with Type 1 or Type 2 Diabetes below to see efficacy and safety results from the 4 EDITION clinical studies
-
EDITION 11,3T2DM: Previously on basal and mealtime insulin ± metformin
-
EDITION 21,4T2DM: Previously on basal insulin and OADs
-
EDITION 31,5T2DM: Insulin-naive patients on OADs
-
EDITION 41,6T1DM: Previously on basal and mealtime insulin
EDITION 11,3T2DM: Previously on basal and mealtime insulin ± metformin
Mean A1C change at 26 weeks vs -0.87% for Lantus*
Incidence of severe hypoglycemia
All registered trademarks cited are property of their respective owners.
*Eligibility Restrictions and Offer Terms:
This offer is not valid for prescriptions covered by or submitted for reimbursement under Medicare, Medicaid, VA, DOD, TRICARE, or similar federal or state programs including any state pharmaceutical assistance program. If you have an Affordable Care plan, you may still be qualified to receive and use this savings card. The Federal Employees Health Benefits Program is not a federal or state government health care program for purposes of the savings program. Void where prohibited by law. Upon registration, patients receive all program details. Sanofi US reserves the right to change the maximum cap amount, rescind, revoke or amend the program without notice.
- Pay as low as $0 up to $99 for a 30-day supply, depending on insurance coverage. Valid up to 10 packs per fill Offer valid for one fill per 30-day supply
Eligibility Restrictions and Offer Terms:
Lantus Insulin Vs Novolin N
Diabetes is a condition in which a person’s blood sugar level is higher than normal. Novolin N and Lantus are types of insulin used to keep blood sugar within normal limits. Classification Lantus and Novolin N are classified therapeutically as antidiabetics or hormones. Their pharmacologic classification is pancreatics. Action Both medications lower blood sugar levels by stimulating absorption of glucose in skeletal muscle and fat. They also inhibit the production of glucose in the liver. Onset Novolin N is an intermediate-acting insulin that starts acting in from one to four hours after being taken. Lantus is a long-acting insulin that starts acting in from one to 10 hours after being injected. Peak The peak time of an insulin is the time during which the blood sugar is most affected. The peak time for Novolin N is four to 12 hours after administration. Lantus does not have a peak time because it is released at a constant rate. Duration Novolin N lasts for from 12 to 18 hours, while Lantus has a duration of up to 24 hours.Continue reading > >
Don’t Miss: Metformin Urinary Side Effects
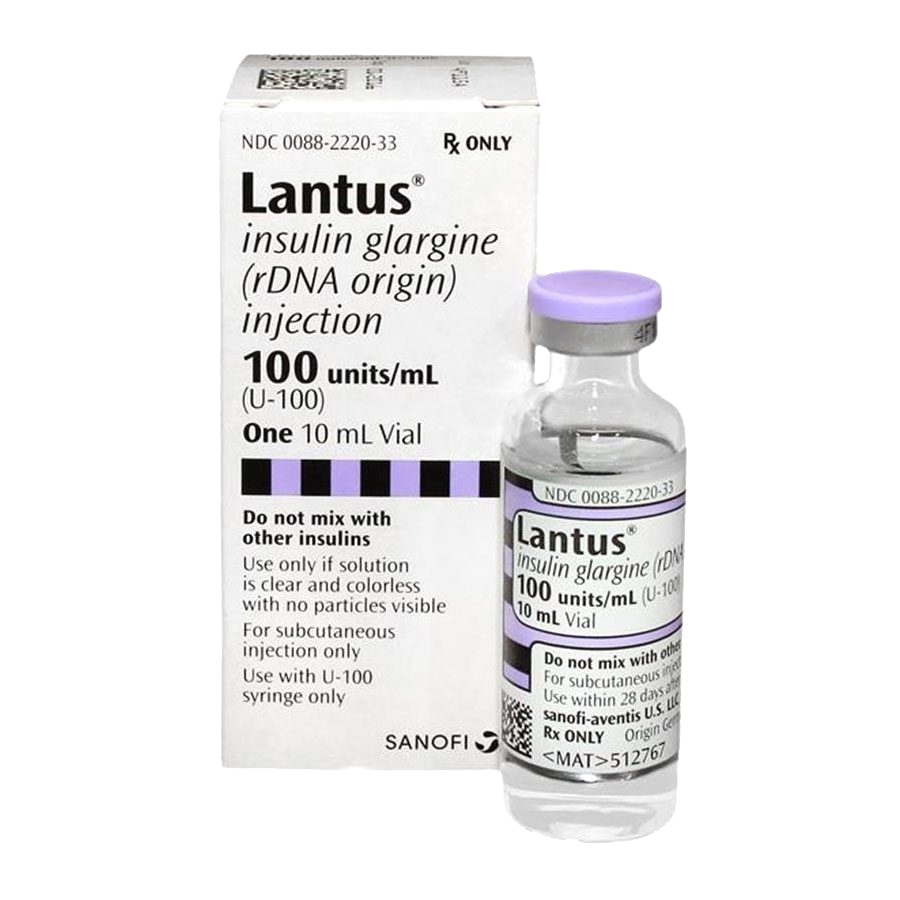
Drug Forms And Strengths
Lantus comes as a liquid solution inside vials and disposable, prefilled pens.
Each vial of Lantus contains 10 milliliters of solution. Each mL contains 100 units of insulin glargine. So, theres a total of 1,000 units of insulin glargine in each vial. This strength is also called Lantus U-100.
The disposable, prefilled pen of Lantus is called SoloStar. Each pen contains 3 mL of solution. There are 100 units of insulin glargine in each mL. That adds up to a total of 300 units of insulin per Lantus SoloStar pen.
Lantus SoloStar pens come in a package of five.
Needles arent included in the packages of either form of Lantus.
The Lantus vial and Lantus SoloStar pen are both meant to be used more than once. The number of times will be different for each person, depending on your dosage. You can use the vials and pens for up to 28 days after you open them. After that time, you should dispose of the vial or pen, even if it still contains some of the drug.
But you should use each needle only once.
Tresiba Vs Lantus: Differences Similarities And Which Is Better For You
Drug overview & main differences | Conditions treated | Efficacy | Insurance coverage and cost comparison | Side effects | Drug interactions | Warnings | FAQ
Diabetes mellitus is a disease that affects over 10% of the United States population, with 1.5 million newly diagnosed cases each year. Diabetes mellitus presents in two forms: Type 1 and Type 2. With Type 1 diabetes, the body does not produce the insulin needed to adequately control blood glucose levels, sometimes called blood sugar. In this instance, insulin injections are the first-line treatment. The body of a Type 2 diabetic makes some insulin but does not utilize it properly. Type 2 is much more prevalent in the United States. In some cases, diet, exercise, and/or oral diabetic medications may adequately control blood sugar. In many cases, however, insulin injections may be necessary to control blood glucose levels for Type 2 diabetics as well.
Tresiba and Lantus are two examples of basal insulin, sometimes called background or long-acting insulin. This type of insulin ensures consistent blood sugar levels throughout the day.
Don’t Miss: What Are The Most Common Side Effects Of Metformin
How Effective Is Tresiba Vs Lantus
Both Lantus and Tresiba are proven to be effective treatments for type 1 and type 2 diabetes, but which is the most effective?
Two reviews of the research into the effectiveness of Tresiba and Lantus found both medications reduced blood glucose by a comparable amount. Both reviews found that Tresiba was less likely to cause hypoglycemia and concluded that it may be more suitable for patients who are prone to hypoglycemia.
Study Design And Patients
ELEMENT 5 was a phase III, prospective, multinational, multicenter, two-arm, active-control, open-label, parallel assignment, randomized controlled trial in patients with T2D, which was conducted from December 2014 to July 2016 in India, Korea, Taiwan, Russia, Turkey, the USA, and Puerto Rico. The primary objective was to determine whether LY IGlar is noninferior to IGlar , as measured by change in glycated hemoglobin A1c over the treatment period. The trial consisted of a screening visit , randomization visit , 24-week treatment period , and a post-treatment-period telephone follow-up visit after approximately 4 weeks . The study was conducted in accordance with the International Conference on Harmonisation Guidelines for Good Clinical Practice and the Declaration of Helsinki . The protocol was approved by the appropriate ethics review boards, and all patients provided written informed consent.
Don’t Miss: Are Cheetos Bad For Diabetics
Are Semglee And Lantus The Same Thing
Semglee and Lantus Injection are long-acting human insulin analogs indicated to improve glycemic controlin adults and pediatric patients with type 1 diabetes mellitus and in adults with type 2 diabetes mellitus.
Side effects of Semglee and Lantus that are similar include low blood sugar , and injection site reactions .
Side effects of Semglee that are different from Lantus include allergic reactions, changes in body fat distribution , itching, rash, fluid retention , and weight gain.
Both Semglee and Lantus may interact with antidiabetic agents, ACE inhibitors, angiotensin II receptor blocking agents, disopyramide, fibrates, fluoxetine, monoamine oxidase inhibitors , pentoxifylline, pramlintide, salicylates, somatostatin analogs , sulfonamide antibiotics, atypical antipsychotics, corticosteroids, danazol, diuretics, estrogens, glucagon, isoniazid, niacin, oral contraceptives, phenothiazines, progestogens , protease inhibitors, somatropin, sympathomimetic agents , thyroid hormones, alcohol, beta-blockers, clonidine, lithium salts, guanethidine, and reserpine.
Lantus may also interact with propoxyphene.
Similar Efficacy And Safety Of Basaglar And Lantus In Patients With Type 2 Diabetes In Age Groups : A Post Hoc Analysis From The Element
To compare efficacy and safety of Basaglar to Lantus in older or younger patients with type 2 diabetes . This subgroup analysis of a phase 3, randomized, double-blind, multinational, 24-week study compared LY IGlar and SA IGlar on several clinical efficacy , basal insulin dose, weight) and safety outcomes in patients either 65 or < 65years. Compared with patients aged < 65years , patients aged 65years had a significantly longer duration of diabetes lower baseline A1c and body weight and body mass index and were more likely to report prestudy SA IGlar use. Compared to patients < 65years, patients 65years needed a lower basal insulin dose and experienced lower body weight gain. There were no significant treatment-by-age interactions for the clinical efficacy and safety outcomes, indicating that there was no differential treatment effect for patients 65years vs those < 65years. Moreover, within each age subgroup, LY IGlar and SA IGlar were similar for all clinical efficacy and safety outcomes. LY IGlar and SA IGlar exhibit similar efficacy and safety in patients with T2D who are 65years and in those < 65years. Eli Lilly and Company and Boehringer-Ingelheim. Plain language summary available for this article. The aim of this phase 3 clinical study was to compare the efficacy and safety of two drugs, Basaglar and Lantus , in patients with type 2 diabetes that were either 65years of age and/or older or younger than 65years of age. This studyContinue reading > >
Don’t Miss: Type 2 Diabetes Genes
Will Basaglar Work As Well As Lantus
In the coming weeks and months, we should hear about how the public responds to Basaglar. However, in the meantime the FDA has said that Basaglar works sufficiently similar to Lantus.
Lillys website states that in a clinical study, patients with inadequately managed type 1 diabetes participated in a 24-week open-label, active-controlled study with a 28 week extension in order to see the effects of Basaglar compared to another insulin glargine product or a non-US-approved insulin glargine product in combination with fast acting insulin lispro at mealtimes.
At 24 weeks treatment with Basaglar resulted in a mean reduction in A1c that was non-inferior to the A1c seen with comparable insulin glargine products.
The worry for many people with diabetes who rely on insulin is that if their pharmacy benefit managers choose Basaglar over Lantus and Basaglar doesnt work as effectively, they may find themselves paying even more than before or entering a fight with their insurance over their preference.
If Basaglar becomes beloved among users, it and other biosimilar insulin coming up behind it have the potential to drive down the cost of insulin.
Have you tried Basaglar? If so, what is your experience with it?
What If I Miss A Dose
If you miss a dose, take it as soon as you remember. If its almost time for your next dose, skip the missed dose and go back to your normal schedule.
Dont take more than one dose in 24 hours unless your doctor tells you to. This could increase your risk for serious side effects, such as hypoglycemia .
Medication reminders can help make sure that you dont miss a dose.
Recommended Reading: How Long Does It Take To Lower Blood Sugar
Is Tresiba Or Lantus More Effective
A meta-analysis of 15 clinical studies published in 2019 looked at data from more than 16,000 participants combined. Tresiba produced a larger decrease in blood sugar levels, but overall, the effects of Tresiba and Lantus on hemoglobin A1C were similar. Tresiba did have a statistically significant less chance of causing hypoglycemia. Researchers concluded that while overall glycemic control was similar, Tresiba may be preferred due to the decreased chance of hypoglycemia.
A meta-analysis published in 2018 had similar findings. Tresiba was significantly less likely to cause hypoglycemia events. For this reason, Tresiba may be preferred over Lantus.
This article is not intended to provide medical advice. Your healthcare provider will decide which types of insulin options are best for you.
What Are The Ingredients In Tresiba And Lantus
Tresibas active drug is insulin degludec. Lantuss active drug is insulin glargine.
Both active drugs are long-acting insulins. This means they work over time to keep your blood sugar levels steady throughout the day, in between meals, and during the night. Tresiba works for up to 42 hours, and Lantus works for up to 24 hours.
Tresiba and Lantus are both prescribed to help people with diabetes manage their blood sugar levels. The lists below give details on the uses of each drug.
- Tresiba and Lantus are both used to:
- manage blood sugar levels in adults with type 1 or type 2 diabetes
Note: Tresiba and Lantus arent approved to treat diabetes ketoacidosis . This is a serious, life threatening complication of diabetes. If you have questions about DKA, talk with your doctor.
Tresiba and Lantus both come as liquid solutions that are available in the following forms:
- Vials. With the vials, you use a new syringe and needle for each dose.
- Prefilled pens. The solution is already inside these pens. You use a new needle for each dose. You discard the pen once the doses are used up . The Tresiba prefilled pens are called FlexTouch, and the Lantus pens are called SoloStar.
Both drugs are given by subcutaneous injection . And theyre both typically used once per day.
You May Like: Does Metformin Cause Hypoglycemia
Basaglar For Type 1 Diabetes
Basaglar is FDA-approved to improve blood sugar levels in adults and children with type 1 diabetes.
If you have type 1 diabetes, your pancreas doesnt make the hormone called insulin. This hormone helps your body move sugar from your blood into your cells. Once sugar has moved into your cells, its processed and used for energy.
Without insulin, the sugar doesnt enter your cells, affecting how well they function. This also leads to high blood sugar levels, which can cause serious problems if not treated.
Type 1 diabetes is a chronic condition. If not treated, it can lead to serious problems like nerve damage. Because insulin is so important for your body to function well, people with this condition need to use insulin-medications in order to survive.
Note: Basaglar shouldnt be used to treat diabetic ketoacidosis , which is a possible complication of diabetes.
Effectiveness for type 1 diabetes
In clinical studies, Basaglar was found effective to treat type 1 diabetes in adults. During studies, peoples response to diabetes treatment is often measured by testing hemoglobin A1c . This measurement shows your average blood sugars over the past 2 to 3 months. The American Diabetes Association recommends a HbA1c goal of less than 7.0% for most adults.
In the study, another group of people with type 1 diabetes took a different insulin glargine medication and a mealtime insulin. These people had similar improvements in their blood sugar levels, as did the people using Basaglar.

