Ceramide Synthesis And Degradation
The rate of ceramides generation depends mainly on the availability of long-chain saturated fatty acids, which participate in the novo ceramide synthesis within the endoplasmic reticulum . Ceramides vary in acyl-chain lengths from C14:0 to C30:0. At the first, rate-limiting step of the de novo pathway, serine palmitoyltransferase initiates the condensation of serine and palmitoyl-CoA to produce 3-ketosphinganine. Further reactions lead to the formation of sphinganine which is acylated to dihydroceramide by six ceramide synthase enzymes , and then to ceramide through dehydroceremide desaturase. Among various acyl-CoAs, palmitoyl-CoA is a highly specific substrate of mammalian SPT in vitro and consequently, in mammals, the chain length of the sphingoid bases is mainly C18 . Recent studies revealed that exogenous fatty acids, cytokines , and even UVB radiation elevate ceramide generation by upregulating the expression of SPT .
Ceramide may also be formed by hydrolysis of sphingomyelin by the neutral and acid sphingomyelinases , which is a stress-activated pathway for ceramide synthesis. These enzymes break down sphingomyelin into ceramide and phosphocholine and are upregulated in response to TNF- , Fas ligand , TLR4 activation , or oxidative stress .
Ceramide can be phosphorylated , rapidly deacylated , or glucosylated . The enzyme sphingomyelin synthase transfer a phosphocholine group from phosphatidylcholine to ceramide, whereby sphingomyelin is reformed .
Implications For The Active Form Of Human Insulin Based On The Structural Convergence Of Highly Active Hormone Analogues
See allHide authors and affiliations
- For correspondence:
Edited* by Donald F. Steiner, University of Chicago, and approved December 15, 2009
Crystallization Structure Determination And Refinement
Crystallization conditions were found initially by an in-house developed screen based on the available insulin crystallization protocols and they were refined individually by the hanging drop method. X-ray data were processed by HKL2000 and all computational crystallography was performed by the CCP4 suite of programs . Structures were determined by molecular replacement with differently truncated 1mso.pdb-derived monomeric insulins as the model . Crystallization details, x-ray data, refinement statistics, and model building protocols are provided in SI Text.
Don’t Miss: How Many Carbohydrates Should A Diabetic Eat A Day
What Does Insulin Do
Insulin is a hormone that is central to regulating glucose metabolism in the body to produce energy. Insulin causes cells in the liver, muscle, and fat tissue to take up glucose from the blood, which it then converts to glycogen which is stored in the liver and muscle. When insulin is absent, glucose is not taken up by body cells and the body begins to use fat as an energy source.
Types Of Insulin Administration With Needles Pumps Pens And Why Insulin Is So Expensive
What does insulin do? Help your body turn food into energy, for starters. When youre diabetic, and youre either not producing insulin, or your insulin function is off, all sorts of things can go wrong. From needles, to pens, to pumps, to types of insulin, were here to empower you with clear answers to all your pressing Qs.
In This Article:
Alvin Powers MD, Mihail Zilbermint MD, and Irl Hirsch MD
Don’t Miss: Sugar Content In Bananas Diabetes
Insulin And Type 1 Diabetes
In type 1 diabetes, the body produces insufficient insulin to regulate blood glucose levels.
Without the presence of insulin, many of the bodys cells cannot take glucose from the blood and therefore the body uses other sources of energy.
Ketones are produced by the liver as an alternative source of energy, however, high levels of the ketones can lead to a dangerous condition called ketoacidosis.
People with type 1 diabetes will need to inject insulin to compensate for their bodys lack of insulin.
Biology: Carb Protein Lipid Or Nucleic Acid
Front Back .Wirisformula Deoxyribonucleic Acid Nucleic Acid Estrogen Lipid Cellulose Carbohydrate Triglycerides Lipids Testosterone Lipid Starch Carbohydrate Glucose Carbohydrate Ribonucleic Acid Nucleic Acid Keratin Protein Fructose Carbohydrate Lactose Carbohydrate Glycogen Carbohydrate Adenosine Triphosphate Nucleic Acid Earwax Lipid Hemoglobin Protein Phospholipids Lipid Sucrose Carbohydrate Steroids Lipids Enzymes Protein Collagen Protein Dr. Axe Collagen Protein – Get 20% Off Today Get 20% Off Today. Just Add Code “Try20” at Checkout. store.draxe.com Related Flashcards Featured Flashcards Related TopicsContinue reading > >
Recommended Reading: What Is A High Blood Sugar Reading
Making Use Of The Differences
The differences between the bacterial and eukaryotic ribosomes are exploited by pharmaceutical chemists to create antibiotics that can destroy a bacterial infection without harming the cells of the infected person. Due to the differences in their structures, the bacterial 70S ribosomes are vulnerable to these antibiotics while the eukaryotic 80S ribosomes are not. Even though mitochondria possess ribosomes similar to the bacterial ones, mitochondria are not affected by these antibiotics because they are surrounded by a double membrane that does not easily admit these antibiotics into the organelle. A noteworthy counterexample, however, includes the antineoplastic antibiotic chloramphenicol, which successfully inhibits bacterial 50S and eukaryotic mitochondrial 50S ribosomes. The same of mitochondria cannot be said of chloroplasts, where antibiotic resistance in ribosomal proteins is a trait to be introduced as a marker in genetic engineering.
Insulin And Fat Storage
As well as being involved in the regulation of blood glucose, insulin is also involved in how fat is used by the body. When the liver is has taken up its capacity of glycoge, insulin signals fat cells to take up glucose to be stored as triglycerides.
An additional effect of insulin is in inhibiting the breakdown of fats.
You May Like: Insulin Antagonist
The Role Of Gpcrs In Circadian Rhythms
New observations have especially intrigued scientists in light of the recently recognized association of sleep disturbances with obesity and risk for type 2 diabetes. Genetic studies have found that a GPCR well known to be involved in sleep rhythms is also important for islet cell function. This GPCR binds to melatonin. Melatonin is best known for its role in sleep and circadian rhythms, but researchers recently learned that individuals who carry a particular mutation in one of the GPCRs that bind to melatonin, melatonin receptor 1B , have higher fasting glucose levels. With that new knowledge in hand, clinical investigators went on to discover that individuals with a mutation in the MTNR1B gene have impaired insulin secretion from their beta cells when glucose levels increase. One manifestation of this is decreased insulin secretion in response to the intake of glucose . Human islets express two different melatonin receptors, 1A and 1B, and the recent recognition of their potential importance to metabolism has increased interest in the role of these GPCRs in alpha and beta cells. While much remains to be learned in this new area of research, it is now clear that circadian rhythms can impact glucose metabolism, in part, through GPCRs expressed in islets.
What Causes Someone To Be Prescribed Insulin
If your body doesnt make insulin or doesnt make enough, you are eventually diagnosed with type 1 diabetes. It used to be called juvenile diabetes, but new estimates show that as many as half of people with type 1 diabetes are not diagnosed until adulthood. On the other hand, if your body doesnt use insulin properly, you have type 2 diabetes.
While people with type 1 diabetes need to take insulin to survive, many people with type 2 are able to stave off insulin use or even avoid it altogether by exercising, losing weight, adapting healthier eating habits, or using other prescription medications.
Recommended Reading: Which Pancreatic Cells Release Insulin And Glucagon
Structural Analysis And Synthesis
Purified animal-sourced insulin was initially the only type of insulin available for experiments and diabetics. John Jacob Abel was the first to produce the crystallised form in 1926. Evidence of the protein nature was first given by Michael Somogyi, Edward A. Doisy, and Philip A. Shaffer in 1924. It was fully proven when Hans Jensen and Earl A. Evans Jr. isolated the amino acids phenylalanine and proline in 1935.
The amino acid structure of insulin was first characterized in 1951 by Frederick Sanger, and the first synthetic insulin was produced simultaneously in the labs of Panayotis Katsoyannis at the University of Pittsburgh and Helmut Zahn at RWTH Aachen University in the mid-1960s.Synthetic crystalline bovine insulin was achieved by Chinese researchers in 1965. The complete 3-dimensional structure of insulin was determined by X-ray crystallography in Dorothy Hodgkin‘s laboratory in 1969.
Two other Nobel Prizes have been awarded for work on insulin. British molecular biologist Frederick Sanger, who determined the primary structure of insulin in 1955, was awarded the 1958 Nobel Prize in Chemistry.Rosalyn Sussman Yalow received the 1977 Nobel Prize in Medicine for the development of the radioimmunoassay for insulin.
Is The Insulin Receptor Considered An Enzyme
Can we consider insulin receptor an enzyme? In other words, does the insulin receptor have enzymatic characteristics?
Yes, the insulin receptor can be viewed as an enzyme, since it catalyzes a reaction — phosphorylation of tyrosine residues on its substrates. But since the protein has multiple functions, it’s probably better to say that the insulin receptor “has enzymatic activity”, rather than “is an enzyme”.
The notion “enzyme” is not restricted to catalysis of reactions involving small molecules, like those in central metabolism. The IUPAC definition of an enzyme reads: “Macromolecules, mostly of protein nature, that function as catalysts by increasing the reaction rates”. So “enzyme” is a broad term, including catalysis of all chemical reactions. DNA replication, mRNA transcription, protein synthesis and posttranslational modifications all involve enzymes. Those enzymes that act on small metabolites can be called “metabolic enzymes” to clarify their function.
Read Also: How Many Points Does Metformin Lower Blood Sugar
Tlk16998 Increases Ir Signaling In 3t3
To explore the effect of TLK16998 on insulin action in cells, differentiated 3T3-L1 adipocytes were treated with the compound. Insulin, at a concentration of 5.6 nmol/l, increased the phosphorylation of the IR -subunit and IRS-1, and the amount of PI 3-kinase that coimmunoprecipitated with IRS-1 . TLK16998 by itself had no significant effect on these parameters. TLK16998, in combination with 5.6 nmol/l insulin, however, caused a greater increase in the phosphorylation of both proteins and in PI 3-kinase coimmunoprecipitation than 5.6 nmol/l insulin alone . The effect of the compound was observable at 3.2 mol/l and was maximal at 10 mol/l. Higher concentrations of TLK16998 were progressively less effective. TLK16998, at concentrations up to 40 mol/l, did not increase the autophosphorylation of the EGF receptor in either the presence or absence of EGF .
Measurement Of Glut4 Distribution
3T3-L1 adipocytes were differentiated as above on a microscope chamber slide. The cells were serum-starved for 16 h and stimulated with TLK16998, insulin, or a combination of the two for 1 h at 37°C. The cells were permeabilized by treating with 3.5% paraformaldehyde , incubated with a mouse anti-GLUT4 antibody for 30 min, washed, incubated with a fluorescein isothiocyanateconjugated anti-mouse antibody for an additional 30 min, washed again, and mounted for examination by confocal microscopy.
You May Like: Which Pancreatic Cells Release Insulin And Glucagon
Faq: Frequently Asked Questions
1. Why is insulin so expensive?
Though reforms are underway in many parts of the US, insulin costs are still prohibitively expensive for many people with diabetes. Reasons include the complexity of the pharmaceutical supply chain and lack of generic substitutes.
2. What is sliding scale insulin?
Sliding scale therapy is a regimen that prescribes a progressive increase in insulin doses before meals and at bedtime, based on your blood sugar levels.
3. What is an insulin index?
The insulin index gives foods a rating based on how much your blood insulin concentration rises in the two hours after consumption.
4. What is an insulin resistance diet?
An insulin resistance diet incorporates foods that will help maintain your bodys balance of insulin and blood sugar. Think nourishing calories from veggies, fruit, lean proteins, and healthy fats.
So What Is A Hormone Really
Hormones are chemical components that are produced in one part of the body and function at an entirely different one . They include proteins, peptides and lipids, existing usually as precursor molecules . In addition hormones have various functions such as the regulation of metabolism and its development. A major role is to act as messengers and carry information around tissues in the body by binding receptors on cell surfaces or within a cell . Receptors are proteins on cell surfaces or within them with available sites for the signalling ligand molecule to bind on. The receptor’s major role is to act as the conductor for hormones and drugs to reach the body and the target they require. One of the main peptide hormones studied through years due to its importance and necessity is insulin, whose structure, synthesis and function are discussed further below.
Don’t Miss: Can You Use Insulin Straight From The Fridge
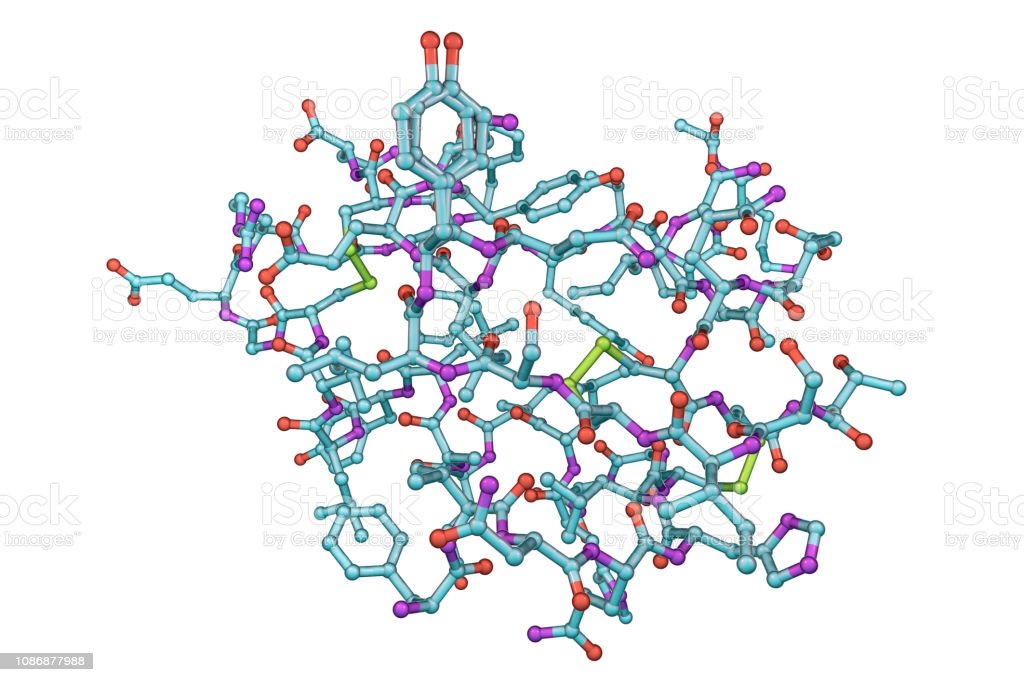
What About Insulin’s Structure It Seems To Be Very Complicated
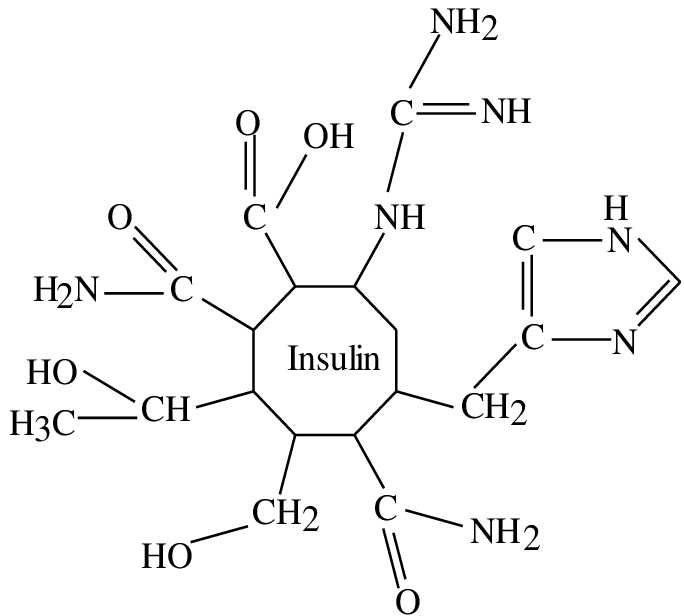
Insulin is a peptide hormone composed of 51 amino acids and has a molecular weight of 5808 Da. It is produced in the islets of Langerhans in the pancreas. The name comes from the Latin insula for “island”. Insulin’s structure varies slightly between species of animals. Insulin from animal sources differs somewhat in “potency” in humans because of those variations. Pig insulin is especially close to the human version. Insulin was the first protein whose amino acid sequence was identified and the first to be synthesized. It is initially synthesized as a precursor molecule known as proinsulin and on cleavage it gives two peptides chains, one comprising of 21 amino acids and the other of 30 amino acids, being mostly conserved in humans and animals . The two chains are joined together by two disulfide bonds between two cysteine residues, where a disulfide bond is the linkage bond between two sulfurs as shown below.
Figure 1. The two chains comprising the insulin structure, A and B, indicating also a disulfide bond between cysteines , since they are very important in stabilizing the two chains together. Chain structure from Ref. .
The insulin hexamer with two of the main binding sites shown next to it. From Ref..
Regulation Of Gene Expression
The activated IRS-1 acts as a secondary messenger within the cell to stimulate the transcription of insulin-regulated genes. First, the protein Grb2 binds the P-Tyr residue of IRS-1 in its SH2 domain. Grb2 is then able to bind SOS, which in turn catalyzes the replacement of bound GDP with GTP on Ras, a G protein. This protein then begins a phosphorylation cascade, culminating in the activation of mitogen-activated protein kinase , which enters the nucleus and phosphorylates various nuclear transcription factors .
Read Also: Metformin Er Max Dose
Does Insulin Make You Hungry
These experiments show that elevations in insulin produce increased hunger, heightened perceived pleasantness of sweet taste, and increased food intake. Finally, a study is described that considers how different insulin levels, produced by the type of food ingested, may affect subsequent food intake.
Biochemistry And Electrophysiology Of Insulin Secretion
Secretion of insulin from -cells is not only an important step in the regulation of glucose homeostasis in healthy individuals, but has also been demonstrated to be impaired in both Type 1 and Type 2 DM . In fact, in the prediabetic state of Type 1 DM as well as in various forms of Type 2 DM, abnormalities in insulin secretion are an integral component of the pathophysiology . In light of its extensive characterization the -cell also serves as a model of the secretory process for other cell types.
Insulin is stored in large dense core vesicles and released by exocytosis as described above. Such release is a multistep process that consists of the transport of the secretory vesicles to the plasma membrane, then docking, priming, and finally fusion of the vesicle with the plasma membrane. This process is regulated cooperatively by nutrients, other hormones, and neurotransmitters in association with electrical depolarization of the -cell and release of insulin. Only a small portion of the insulin stored in vesicles in the -cell is released, however, even under maximum stimulation. This suggests that systemic insulin levels are regulated by secretion rather than by biosynthesis and is not ordinarily limited by the size of storage pools. However the mechanisms that regulate the directed transport of the insulin granules to the plasma membrane are also not well understood.
Don’t Miss: How Long Do Diabetics Live
Insulin Amino Acid Sequence
The primary amino acid sequence of human insulin is detailed in both panels of Figure 6-4A/B.
Figure 6-4A. Amino acid sequence and structure of insulin and human preproinsulin. Amino acid sequence of preproinsulin.
The preproinsulin has 110 amino acids. Dashed circles around either an R or K indicate sites of peptidase cleavage that result in generation of the mature insulin that is comprised by two separate peptides: the A chain with 21 and the B chain with 30 amino acids. A third C peptide with 21 amino acids served as a linker between the A and B chains. The solid black circles represent mutations which result in a disruption of the formation of disulfide-bond formation and/or the proinsulins normal folding these mutations can lead to the onset of neonatal diabetes. There are also other single amino acid changes that lead to a variety of other insulin-dependent disorders that have clinical consequences.
J Biol Chem246
Figure 6-4B. Three-dimensional orientation of the insulin structure.
Proc.Nat.Acad Sci.
Figure 6-4A shows the amino acid sequence of the human preproinsulin with its 24 amino acid signal peptide attached . The amino acid residues shown in black circles are a sample of genetic mutations that disrupt either the folding of the proinsulin protein or disulfide bond formation. Either of these outcomes can lead to an appearance of the genetic disease known as permanent neonatal diabetes mellitus .
Pran Kishore Deb, … Rakesh K. Tekade, in, 2019

