Insulin And Type 2 Diabetes
Type 2 diabetes is characterised by the body not responding effectively to insulin. This is termed insulin resistance. As a result the body is less able to take up glucose from the blood. In the earlier stages of type 2 diabetes, the body responds by producing more insulin than it would normally need to.
If type 2 diabetes develops over a number of years, the extra demands on the pancreas to produce insulin can lead to a loss of insulin producing cells as they wear out.
Depending on their level of insulin resistance, people with type 2 diabetes may also need to take insulin injections to manage their blood sugar levels.
Diabetes And Potassium Levels
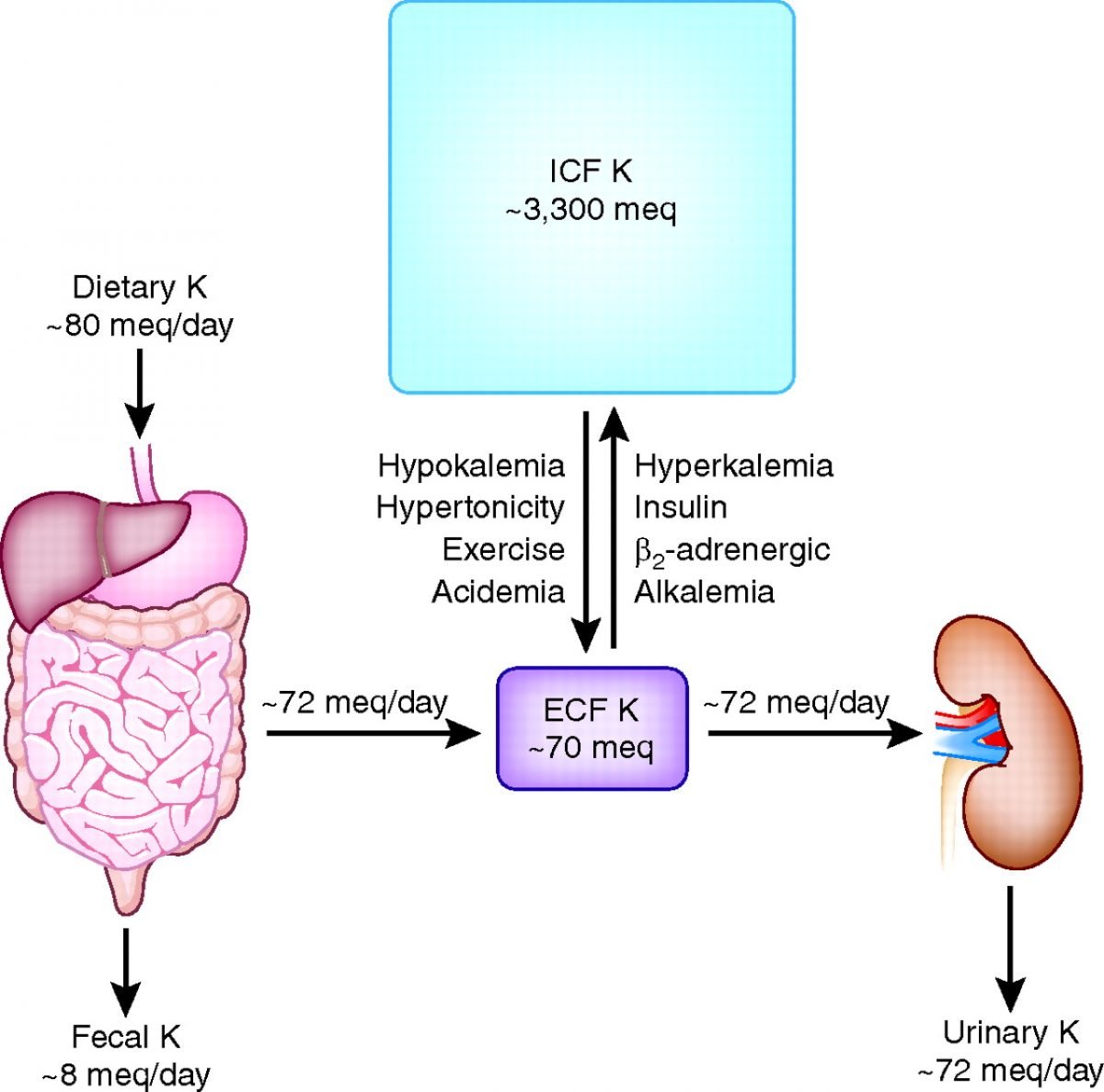
Potassium is a mineral that is often referred to as an electrolyte. These are electrically charged particles that are used by the cells to maintain voltage across the cell membranes of the body. They carry nerve impulses to other cells. Some other electrolytes in the body are chloride, sodium, calcium, and magnesium. Kidneys help in regulating the number of electrolytes in the body.
The role of potassium is to help in nerve conduction, to help in contraction of the muscles, and to help regulate the heartbeat. Potassium also helps to maintain a proper fluid balance between the bodys cells and body liquids. One of the roles of the kidney is to ensure the bodys potassium level is correctly maintained. Levels that are too high or low can be equally dangerous.
The Problem With High Potassium
Too much potassium is called hyperkalemia. It can be a problem in people with poorly controlled diabetes.
High blood sugar damages the kidneys, which normally remove extra potassium from your body. People with diabetes and high potassium are more likely to have heart problems and other complications.
Your doctor might suggest tips like these to lower your potassium if it’s too high:
- Eat a low-potassium diet.
Diabetes in Control: “Hyperkalemia Associated with Poor Outcomes for Diabetes Patients.”
Diabetic Medicine: “Hyperkalaemia in people with diabetes: occurrence, risk factors and outcomes in a Danish population-based cohort study.”
Expert Review of Endocrinology & Metabolism: “Potassium and risk of type 2 diabetes.”
Heart and Stroke Foundation of Canada: “Aldosterone Antagonists.”
Journal of Physiology & Biochemistry: “The role of dietary potassium in hypertension and diabetes.”
Lab Tests Online: “Potassium.”
Mayo Clinic: “Angiotensin-converting enzyme inhibitors,” “Diabetic ketoacidosis: Symptoms & causes,” “Diabetic nephropathy: Symptoms & causes,” “Diuretics: A cause of low potassium?”
National Kidney Foundation: “Six Steps to Controlling High Potassium.”
National Institute of Diabetes and Digestive and Kidney Diseases: “Symptoms & Causes of Diabetes,” “What is Diabetes?”
NIH Office of Dietary Supplements: “Potassium.”
UCSF Health: “Potassium test.”
You May Like: What Happens When You Have Diabetes
The Link Between Potassium And Type 2 Diabetes
Insulin is a hormone produced by the pancreas, and its responsible for regulating blood sugar levels by transporting glucose to the cells in the body. The cells then use glucose for energy, or store it for later use.
Potassium is generally stored in the fluid inside of the cells, but when theres too much glucose outside of the cells , potassium moves outside of the cell, raising potassium levels in the blood.
Insulin then comes to move glucose into the cell to restore potassium homeostasis, causing potassium levels to drop.
When the body either doesnt produce enough insulin, doesnt use insulin properly or is insulin resistant, this causes a buildup of glucose in the bloodstream, since glucose isnt able to enter the cells without insulin.
People with low potassium levels will release less insulin, which causes higher blood sugar levels, and increases the risk of developing type 2 diabetes. When administering insulin, its crucial for healthcare providers to monitor potassium levels.
Relationship Of Insulin To Potassium
Insulin is the hormone that is secreted by the beta cells of the pancreas. It keeps the level of potassium within the normal range in the blood. When insulin decreases the potassium level increases.
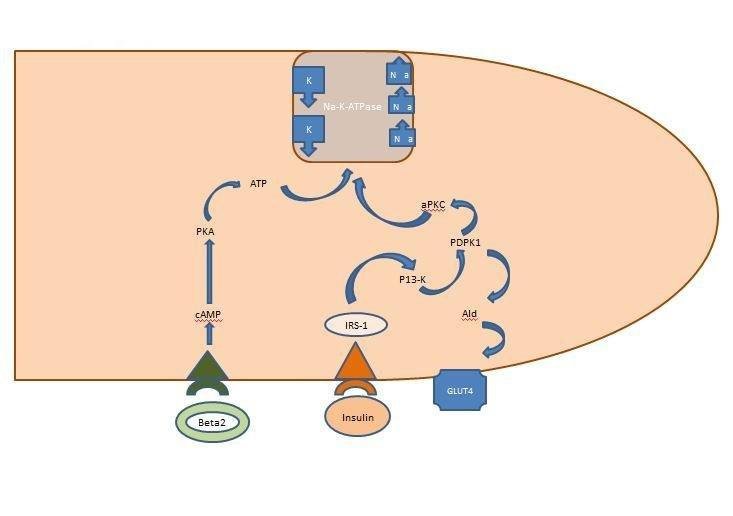
Insulin has plenty of functionsin the body besides lowering blood sugar. One of its functions is to put potassium into the cells by activating the cellular channel of sodium and potassium. Insulin stimulates the uptake of potassium and glucose in all the bodys cells, but primarily it fuels the fat cells as well as muscle cells.
In type 2 diabetes insulin does not function properly. Therefore, the body cells become resistant to insulin and blood glucose levels are elevated.
Recommended Reading: Is 500 Blood Sugar Dangerous
Physiologic Effects Of Insulin
Stand on a streetcorner and ask people if they know what insulin is, and many will reply, “Doesn’t it have something to do with blood sugar?” Indeed, that is correct, but such a response is a bit like saying “Mozart? Wasn’t he some kind of a musician?”
Insulin is a key player in the control of intermediary metabolism, and the big picture is that it organizes the use of fuels for either storage or oxidation. Through these activities, insulin has profound effects on both carbohydrate and lipid metabolism, and significant influences on protein and mineral metabolism. Consequently, derangements in insulin signalling have widespread and devastating effects on many organs and tissues.
The Insulin Receptor and Mechanism of Action
Like the receptors for other protein hormones, the receptor for insulin is embedded in the plasma membrane. The insulin receptor is composed of two alpha subunits and two beta subunits linked by disulfide bonds. The alpha chains are entirely extracellular and house insulin binding domains, while the linked beta chains penetrate through the plasma membrane.
Insulin and Carbohydrate Metabolism
It should be noted here that there are some tissues that do not require insulin for efficient uptake of glucose: important examples are brain and the liver. This is because these cells don’t use GLUT4 for importing glucose, but rather, another transporter that is not insulin-dependent.
Insulin and Lipid Metabolism
Other Notable Effects of Insulin
Affinity Purification With Bbr
BBR-immobilized beads were equilibrated with 100mM KCl buffer containing 20mM HEPES-NaOH , 100mM KCl, 1mM MgCl2, 0.2mM CaCl2, 0.2mM EDTA,10% glycerol, 0.1% NP-40, 1mM DTT, and 0.2mM PMSF. Cell extracts were prepared from transfected human 293T cells and rat INS-1 -cells as described and were incubated with the beads for 4h at 4°C. The beads were washed three times with 100mM KCl buffer, and bound proteins were eluted with 1× loading dye solution containing 62.5mM Tris-HCl , 0.005% bromophenol blue, 2% SDS, 10% glycerol, and 5% 2-mercaptoethanol. In some experiments, 1mM BBR was added to cell extracts before the incubation with the beads .
Recommended Reading: What Kind Of Rice Can A Diabetic Eat
Bbr Binds To The Kcnh6 Channel Protein On Pancreatic Islet
We used high-performance affinity beads that allowed the purification of drug-targeted proteins from cell extracts to determine whether BBR targeted the KCNH6 protein on pancreatic islet -cells. The active BBR derivative berberrubine was covalently conjugated to the beads . Pull-down experiments were performed using naked or BBR-immobilized beads . Fractions eluted from the beads were subjected to immunoblotting and probed with a KCNH6-specific antibody. KCNH6 was clearly isolated as a BBR-specific binding protein in INS-1 -cells .
KCNH6-null human embryonic kidney 293T cells were transfected with plasmids encoding FLAG-tagged human KCNH6 to confirm the identity of the eluted protein. Cell extracts were isolated, and the pull-down experiments were repeated in the presence or absence of competing BBR in the solution . In the presence of competing BBR, the potential target protein bound to the BBR in the solution, and the amount of target protein binding to the beads would be reduced. When competing BBR was added to the reaction system, the yields of the KCNH6 were significantly reduced compared to those observed in the absence of BBR . These results convincingly show that BBR specifically and directly interacts with the KCNH6 protein molecules.
Hyperinsulinemic Euglycemic Insulin Clamp Technique
On day 9, breakfast was withheld and subjects underwent hyperinsulinemic euglycemic clamp starting at 8:00 a.m. with insulin infusion at 80 mU/m2 of body surface area for 2 hours. A 20-g/dl glucose solution was started after 4 minutes of insulin infusion to maintain plasma glucose concentration at the fasting levels throughout the clamp procedure. Plasma insulin was determined by a modification of the method of Yallow and Berson . Blood for plasma glucose levels was drawn every 5 minutes from an arterialized dorsal hand vein kept in a hotbox at 70°C . A glucose analyzer was used to measure plasma glucose, and the rate of the glucose infusion was adjusted to maintain euglycemia. Peripheral venous blood was drawn without stasis from an antecubital vein for electrolytes and lipid profile at 8:00 and 10:00 a.m. Two timed urine specimens were collected between 6:00 and 8:00 a.m. and 8:00 and 10:00 a.m. . To ensure adequate urinary output, 250 ml of water was given orally at 6:00 and 8:00 a.m. Urine was collected under mineral oil and kept refrigerated until analysis. Plasma and urinary electrolytes and plasma lipid profiles were measured by an autoanalyzer .
Don’t Miss: Diabetic Orthostatic Hypotension
Causes Of High Potassium Levels
High potassium levels often result from kidney damage. Kidney damage is usually due to poorly controlled diabetes, and is considered a major complication of diabetes . High potassium can also occur if someone has had diabetic ketoacidosis , a serious metabolic condition more commonly seen in people with Type 1 diabetes. Heart attack, injuries, infections, overuse of potassium supplements, and use of ACE inhibitor drugs may also lead to high potassium levels. Too much potassium, by the way, can lead to weakness, paralysis, irregular heartbeat, or even a heart attack.
Hypokalemia Due To Shift Of Potassium Into Stores
Shift of potassium into stores may cause a rapid reduction in serum potassium concentration to below 3.5 mmol/L. This may result from stimulation of the activity of Na+/K+ pumps in skeletal muscles. Thus, potassium is transferred from plasma to skeletal muscle cells. This type of hypokalemia can arise and vanish within seconds to minutes due to the huge capacity of skeletal muscle Na+/K+ pumps and K+ channels. Because it occurs without any potassium depletion, it is sometimes called pseudohypokalemia to distinguish it from hypokalemia associated with potassium depletion. In cardiac patients, this hypokalemic effect of beta-adrenoceptor agonists as well as its inhibition by beta-adrenoceptor antagonists is of major interest. Also, insulins hypokalemic effect is of interest because these patients often have diabetes as a comorbidity .
The mechanisms involved in the hypokalemic effects of beta-adrenoceptor agonists and insulin are well described. Thus, beta-adrenoceptor agonists stimulate Na+/K+ pump-mediated potassium uptake in skeletal muscles of experimental animals and humans . Likewise, insulin has been shown to increase skeletal muscle Na+/K+ pump-mediated potassium uptake . Interestingly, the combination of albuterol and insulin in patients with renal insufficiency showed an additive hypokalemic effect . On the other hand, another study showed that protection against insulin induced a further reduction in plasma potassium concentration in hypokalemia .
You May Like: Increasing Metformin Dosage Side Effects
Cell Culture And Transfection
All cells were cultured in a humidified incubator with 95% air and 5% CO2 at 37°C. INS-1 832/13 -cells were cultured in RPMI 1640 medium containing 10% FBS, 1mM L-glutamine, 1mM HEPES, 1mM sodium pyruvate, 50M 2-mercaptoethanol, and 1% penicillinstreptomycin. HEK293T cells were cultured in DMEM containing 10% FBS and 1% penicillinstreptomycin. Pancreatic islets and primary pancreatic islet -cells were cultured in RPMI 1640 medium supplemented with 10% FBS and 1% penicillinstreptomycin. Pancreatic islets were isolated from mice euthanized by cervical dislocation through the injection of 500 units/mL collagenase solution into the pancreatic duct, followed by digestion at 37°C for 25min with mild shaking, and isolated islets were picked by hand selection under a dissecting microscope, as described previously,. After isolation and recovery, pancreatic islets were treated with 0.05% trypsin in PBS for 5min at 37°C and fully dispersed to isolate primary pancreatic islet -cells. Kcnh6 knockdown INS-1 -cells were transfected with the Kcnh6 shRNA or scrambled shRNA using Lipofectamine 2000 . The following target sequence was used: 3-ACACGCAGATGCTGCGTGTCAAGGAGTTC-5. The WT KCNH6 cDNA was subcloned into pcDNA3-FLAG and GV314-EGFP vector as previously described. HEK293T cells were transiently transfected using Lipofectamine 2000 reagent according to the manufacturers protocol.
Evolution And Species Distribution
Insulin may have originated more than a billion years ago. The molecular origins of insulin go at least as far back as the simplest unicellular eukaryotes. Apart from animals, insulin-like proteins are also known to exist in the Fungi and Protista kingdoms.
Insulin is produced by beta cells of the pancreatic islets in most vertebrates and by the Brockmann body in some teleost fish.Cone snailsConus geographus and Conus tulipa, venomous sea snails that hunt small fish, use modified forms of insulin in their venom cocktails. The insulin toxin, closer in structure to fishes’ than to snails’ native insulin, slows down the prey fishes by lowering their blood glucose levels.
Don’t Miss: Hypertriglyceridemia Low Carbohydrate Diet
Bbr Does Not Promote Insulin Secretion In The
Fig. 3: BBR targets KCNH6 channel proteins on islet -cells.
a The active derivative of BBR, berberrubine, was immobilized on high-performance affinity beads. b Schematic of the experimental design. BBR-binding proteins were purified from cell extracts using different beads, as indicated. Naked beads were used as a control BBR-immobilized beads were used to pull down BBR-binding proteins in the eluate 1mM BBR was added to the extracts before the incubation with BBR-immobilized beads to reduce the yield of specific binding proteins the same amount of vehicle was added to cell extracts before the incubation with the beads as a control for . Bound proteins were eluted and subjected to western blot analyses. c BBR-binding proteins from INS-1 -cell extracts were analyzed with western blotting using an anti-KCNH6 antibody. A representative immunoblot from three different experiments is shown. d KCNH6-null human embryonic kidney 293T cells were transfected with plasmids encoding FLAG-tagged human KCNH6. BBR-binding proteins from transfected HEK293T cell extracts were analyzed with western blotting using an anti-FLAG antibody. A representative immunoblot from three different experiments is shown.
Kcnh6 knockdown INS-1 -cells were generated with a specific shRNA , and GSIS was evaluated in the KD and the control WT INS-1 cells. Although BBR significantly increased GSIS in the WT INS-1 cells, no differences were noted between BBR- and vehicle-treated KD INS-1 cells .
Insulin And Fat Storage
As well as being involved in the regulation of blood glucose, insulin is also involved in how fat is used by the body. When the liver is has taken up its capacity of glycoge, insulin signals fat cells to take up glucose to be stored as triglycerides.
An additional effect of insulin is in inhibiting the breakdown of fats.
Also Check: Is Blood Sugar Regulated By Positive Feedback
Insulin For The Treatment Of Hyperkalemia: A Double
Keywords: CopyrightHypoglycemia in the treatment of hyperkalemia with insulin in patients with end-stage renal diseasecited by
Potassium plays a critical role in cellular metabolism and normal neuromuscular function. Tightly regulated homeostatic mechanisms have developed in the process of evolution to provide primary defense against the threats of hyper- and hypokalemia. The kidney plays a primary role in potassium balance, by increasing or decreasing the rate of potassium excretion. Distribution of potassium between the intracellular and the extracellular fluid compartments is regulated by physiologic factors such as insulin and catecholamines which stimulate the activity of the Na+-K+ ATPase. Only about 10% of the ingested potassium is excreted via the gut under normal physiologic conditions .
The definition of hypoglycemia has been a topic of debate. The workgroup of the American Diabetes Association and the Endocrine Society defines iatrogenic hypoglycemia in patients with diabetes mellitus as all episodes of an abnormally low plasma glucose concentration that expose the individual to potential harm. A plasma glucose concentration of 70 mg/dL is recommended as the alert value even though symptoms of hypoglycemia usually develop at a level below this threshold . This value allows time for close monitoring of the patient to prevent symptomatic hypoglycemia and has been used to define hypoglycemia in numerous clinical trials.
Disclosures. None declared.
Causes Of Low Potassium Levels
Low potassium levels may be due to something as relatively simple as dehydration from excessive sweating, vomiting, or diarrhea, or to something more serious such as an adrenal gland problem, cystic fibrosis, or severe burns, for example. Low levels can also result from taking diuretics or from malnutrition.
Read Also: Metformin Cause Hypoglycemia
Diabetes Risk And Potassium
Researchers have concluded low potassium blood levels are a risk factor for diabetes.
For example, one study followed 12,209 participants for nine years. Those with lower potassium levels were nearly 1.5 times more likely to develop diabetes, compared to those with higher levels of potassium.
Another studyfollowed 4,409 men and found low blood potassium levels to be predictive of type 2 diabetes, in otherwise healthy men.
It has been proven that African Americans are at increased risk of developing diabetes, and some research has connected this increased risk to potassium levels.
In fact, a 2011 observational study found potassium levels to be lower in African Americans compared to caucasians at baseline. African Americans with potassium levels less than 4.0 mEq/L were 2.28 times more likely to develop diabetes compared to those with normal levels.
Some researcher have proposed that a type of blood pressure medication called Thiazides may increase risk of diabetes due to the fact that Thiazides lower potassium levels.
This study found that each 0.5 mEq/L decrease in potassium increased risk of developing Thiazide-induced diabetes by 45%. Researchers noted Thiazide-induced diabetes typically occurred within the first year of starting Thiazide treatment.
Signs And Symptoms Of Hyperkalemia
Patients with hyperkalemia may be asymptomatic, or they may report the following symptoms :
-
Generalized fatigue
-
Paralysis
-
Palpitations
Evaluation of vital signs is essential for determining the patients hemodynamic stability and the presence of cardiac arrhythmias related to hyperkalemia. Additional important components of the physical exam may include the following:
-
Cardiac examination may reveal extrasystoles, pauses, or bradycardia
-
Neurologic examination may reveal diminished deep tendon reflexes or decreased motor strength
-
In rare cases, muscular paralysis and hypoventilation may be observed
-
Signs of renal failure, such as edema, skin changes, and dialysis sites, may be present
-
Signs of trauma may indicate that the patient has rhabdomyolysis, which is one cause of hyperkalemia
See Clinical Presentation for more detail.
Read Also: Side Effects Of Diabetes Medicine

