Mechanisms Of Insulin Secretion
Increased levels of glucose induce the first phase of glucose-mediated insulin secretion by release of insulin from secretory granules in the cell. Glucose entry into the cell is sensed by glucokinase, which phosphorylates glucose to glucose-6-phosphate , generating ATP. Closure of K+-ATP-dependent channels results in membrane depolarization and activation of voltage dependent calcium channels leading to an increase in intracellular calcium concentration this triggers pulsatile insulin secretion. Augmentation of this response occurs by both a K+-ATP channel-independent Ca2+-dependent pathway and K+-ATP channel-independent Ca2+-independent pathways of glucose action. Other mediators of insulin release include activation of phospholipases and protein kinase C and by stimulation of adenylyl cyclase activity and activation of cell protein kinase A, which potentiates insulin secretion. This latter mechanism may be activated by hormones, such as vasoactive intestinal peptide , PACAP, GLP-1, and GIP. These factors appear to play a significant role in the second phase of glucose mediated insulin secretion, after refilling of secretory granules translocated from reserve pools.
How Are Various Metabolic Processes Interrelated
A metabolic pathway is a step-by-step series of interconnected biochemical reactions that convert a substrate molecule or molecules through a series of metabolic intermediates, eventually yielding a final product or products. For example, one metabolic pathway for carbohydrates breaks large molecules down into glucose.
Sleep And Sleep Deprivation
Acute sleep deprivation in healthy young adults has been reported to raise fasting blood glucose concentrations in association with altered diurnal cortisol secretion and reduced heart rate variability. These effects suggest increased counter-regulatory hormone secretion via hyper-arousal with activation of the hypothalamo-pituitary adrenal axis. There is also accumulating evidence that chronic sleep deprivation may impact on insulin and insulin resistance. Recent epidemiological studies report that reduced sleep duration is associated with increased BMI. Sleep deprivation is associated with decreased plasma concentrations of leptin, the adipocyte peptide hormone regulating fat mass and appetite, and increased concentrations of ghrelin, which increases appetite. Growth hormone is secreted during slow wave sleep, sleep declines with age and growth hormone deficiency in adults has been associated with central adiposity and insulin resistance, but whether sleep deprivation acts through these mechanisms is not clearly established. Obstructive sleep apnoea , where sleep disturbance results from obstruction to breathing during sleep, is associated with impaired glucose tolerance independent of adiposity, and improves with continuous positive airway pressure treatment but whether this is due to resolution of hypoxia and hypercapnia, or to effects on sleep quality, is unclear.
Recommended Reading: Metformin Meaning
Insulin Secretion In Response To Stimuli
Response to Glucose
In healthy individuals glucose stimulated pancreatic secretion is biphasic. Intravenous administration of glucose is associated with a rapid first phase of insulin release within 1 minute, peaking at 35 minutes, and lasting about 10 minutes the slower onset second phase of insulin secretion begins shortly after the glucose bolus but is not apparent until 10 minutes later, lasts the duration of the hyperglycaemia and is proportional to the glucose concentration immediately prior to the glucose administration. The first phase of insulin secretion represents release of insulin already synthesised and stored in secretory granules the second phase represents secretion of both stored and newly synthesised insulin. Overall insulin secretion relates to the total dose of glucose and its rate of administration maximal pancreatic response occurs with 20 g of glucose given intravenously over 3 minutes in humans.
In contrast to the reproducible pattern of insulin secretion in response to intravenous glucose, insulin secretion following oral glucose is much more variable. With an oral glucose load, gastric emptying and gastrointestinal motility affect glucose absorption, gastro-intestinal hormones and neural input associated with glucose ingestion modify the insulin response, and insulin secretion continues some time after glucose ingestion.
Response to Arginine
Effects of Lipids
Response to Mixed Meal
Incretin Hormones
Effects of Neural and Hormonal Stimuli
Physiology Of Insulin Secretion
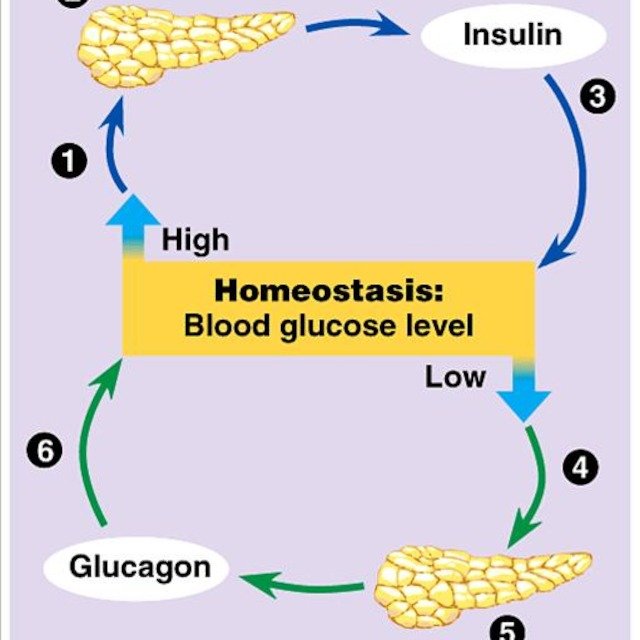
Glucose is the principal stimulus for insulin secretion, though other macronutrients, hormones, humoral factors and neural input may modify this response. Insulin, together with its principal counter-regulatory hormone glucagon, regulates blood glucose concentrations. Pancreatic cells secrete 0.251.5 units of insulin per hour during the fasting state, sufficient to enable glucose insulin-dependent entry into cells. This level prevents uncontrolled hydrolysis of triglycerides and limits gluconeogenesis, thereby maintaining normal fasting blood glucose levels. Basal insulin secretion accounts for over 50% of total 24 hour insulin secretion. Following secretion of insulin into the portal venous system, 60% is subsequently removed by the liver so portal vein insulin concentrations reaching the liver approach triple that of the peripheral circulation. In healthy lean individuals circulating venous fasting insulin concentrations are about 315 mIU/L or 1890 pmol/L. Meal-related insulin secretion accounts for the remaining fraction of the total daily output.
Read Also: Maximum Metformin Dose
Do You Have Insulin Resistance
How do you find out if youre insulin resistant? No one test will tell you, but if you have high blood sugar levels, high triglycerides , high LDL cholesterol, and low HDL cholesterol, your health care provider may determine you have insulin resistance.
Important note: Type 1 diabetes is different its thought to be caused by an autoimmune reaction . People with type 1 diabetes dont make enough insulin and need to take it to survive.
Insulin Resistance And Dyslipidemia
The dyslipidemia induced by insulin resistance and type 2 diabetes is characterized by the lipid triad: high levels of plasma triglycerides, low levels of HDL, and the appearance of small dense low-density lipoproteins , as well as an excessive postprandial lipemia . Hypertriglyceridemia increases the incidence of CVD by 32% in men and 76% in women . A study conducted in 10,038 people with normal blood pressure or pre-hypertension demonstrated dyslipidemia as a strong predictor of development of type 2 diabetes . Frequently, diabetic dyslipidemia precedes type 2 diabetes by several years, suggesting that the abnormal lipid metabolism is an early event in the development of CVD in type 2 diabetes .
Obesity is a world-wide epidemic and intimately associated with the development of type 2 diabetes and CVDs. Visceral and epicardial adiposity related to obesity are the major drivers for cardiac disease in these individuals . Obesity has a major effect in modifying the lipoprotein profile and factors associated with systemic and vascular inflammation, and endothelial dysfunction . Abnormal concentrations of lipids and apolipoproteins can produce changes in the production, conversion, or catabolism of lipoprotein particles. These changes may contribute to increased basal lipolysis in obesity and the release of fatty acids into the circulation that consequences a proatherogenic phenotype .
Recommended Reading: Are Bananas Good For Type 2 Diabetes
Insulin Blood Sugar And Type 2 Diabetes
Insulin is a key player in developing type 2 diabetes. This vital hormoneyou cant survive without itregulates blood sugar in the body, a very complicated process. Here are the high points:
- The food you eat is broken down into blood sugar.
- Blood sugar enters your bloodstream, which signals the pancreas to release insulin.
- Insulin helps blood sugar enter the bodys cells so it can be used for energy.
- Insulin also signals the liver to store blood sugar for later use.
- Blood sugar enters cells, and levels in the bloodstream decrease, signaling insulin to decrease too.
- Lower insulin levels alert the liver to release stored blood sugar so energy is always available, even if you havent eaten for a while.
Thats when everything works smoothly. But this finely tuned system can quickly get out of whack, as follows:
- A lot of blood sugar enters the bloodstream.
- The pancreas pumps out more insulin to get blood sugar into cells.
- Over time, cells stop responding to all that insulintheyve become insulin resistant.
- The pancreas keeps making more insulin to try to make cells respond.
- Eventually, the pancreas cant keep up, and blood sugar keeps rising.
Which Is A Common Intermediate Of All Metabolic Pathways
Central Themes of Metabolic Pathways Acetyl CoA is a common intermediate of all metabolic pathways. It interconnects glucose, fatty acid and amino acid metabolism. Oxidation of dietary fuel leads to the capture of energy in the form of ATP and NADH / FADH2. NADH / FADH2 transfer their electrons to O2 via the electron transport chain.
Read Also: Are You Born With Type 2 Diabetes
Structure And Chemical Properties Of Insulin
Insulin was found to be a polypeptide in 1928 with its amino acid sequence identified in 1952. It is in fact a dipeptide, containing A and B chains respectively, linked by disulphide bridges, and containing 51 amino acids, with a molecular weight of 5802. Its iso-electric point is pH 5.5. The A chain comprises 21 amino acids and the B chain 30 amino acids. The A chain has an N-terminal helix linked to an anti-parallel C-terminal helix the B chain has a central helical segment. The two chains are joined by 2 disulphide bonds, which join the N- and C-terminal helices of the A chain to the central helix of the B chain. In pro-insulin, a connecting peptide links the N-terminus of the A chain to the C-terminus of the B chain.
How Are Glucose And Atp Used In Living Things
Almost all living organisms need glucose and ATP for energy. Glucose is utilized to store and transmit energy, whereas ATP powers life activities within cells. Many autotrophs obtain food by photosynthesis, a process in which light energy from the sun is converted into chemical energy that is stored in glucose. Most heterotrophs, including humans, use glucose as their source of energy. During respiration, glucose converts oxygen into carbon dioxide and water while releasing energy that allows organisms to grow and reproduce.
In addition to photosynthesis and respiration, glucose can also be used as a source of energy through the process of glycolysis. This metabolic pathway converts glucose into pyruvate, which can then be oxidized to produce energy or transformed into glycogen for storage. Glycogen can later be converted back into glucose when needed.
All living organisms require energy to live their lives. Autotrophs get this energy from the sun via photosynthesis, while heterotrophs such as humans get it by eating plants or other animals.
Once inside a plant cell, glucose passes through several stages along its path to energy production. First, it is phosphorylated to form glucose-6-phosphate. Next, glucose-6-phosphate is converted into fructose-6-phosphate by the action of hexokinase.
Also Check: Which Statement Is Correct Regarding Glargine Insulin
Common Conditions Associated With Insulin Resistance
Type 2 Diabetes
Following pioneering work by Bornstein and the Nobel Prize-winning work of Yalow and Berson, the first insulin assays became widely available in the late 1960s it was subsequently confirmed that diabetic patients with so-called or maturity onset or type 2 diabetes had normal or increased plasma insulin levels. Insulin resistance was reported to be a characteristic feature of T2DM in the early 1970s. A progressive inability of the cells to compensate for the prevailing insulin resistance by sufficient hyperinsulinaemia, heralds the clinical onset of this disorder. While twin studies and linkage analyses are consistent with a strong genetic component in the development of type 2 diabetes, several decades of research have failed to identify a predominant genetic abnormality in the majority of cases. The aetiology of T2DM is thought to be polygenic, with environmental factors being superimposed upon this basic predisposition.
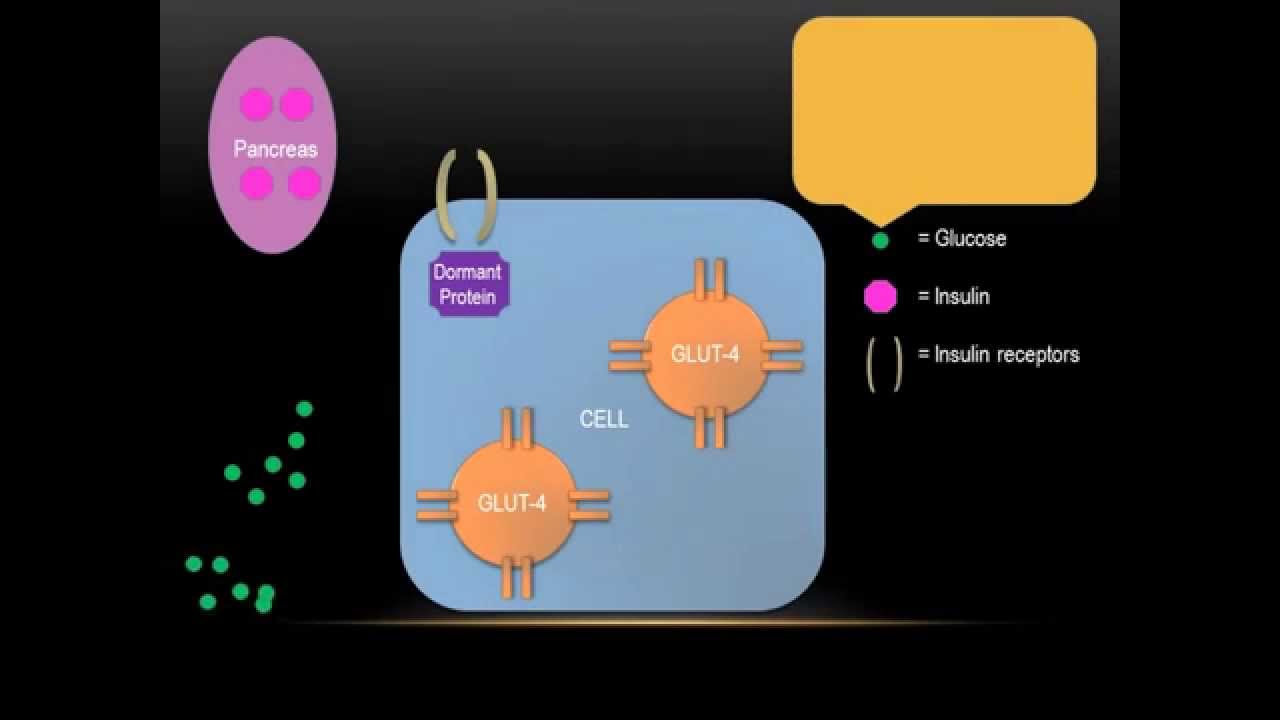
Insulin resistance typically predates the development of diabetes and is commonly found in unaffected first-degree relatives. The morbidity of the disorder relates both to the severity of hyperglycaemia and the metabolic consequences of insulin resistance itself. The primary defects in insulin action appear to be in muscle cells and adipocytes, with impaired GLUT 4 translocation resulting in impaired insulin-mediated glucose transport.
Metabolic Syndrome
-
Abdominal obesity
Men < 40 mg/dL
Women < 50 mg/dL
Dyslipidaemia
Hypertension
PCOS
OSA
The Discovery Of Insulin
In 1889 German scientists Minkowski and von Mering noted, from their experimental work with animals, that total pancreatectomy led to the development of severe diabetes. They hypothesised that a substance secreted by the pancreas was responsible for metabolic control. Others later refined this hypothesis, noting diabetes to be associated with destruction of the islets of Langerhans. While Minkowski, as well as Zuelzer in Germany and Scott in the USA attempted, with inconsistent results, to isolate and administer the missing pancreatic islet substance, Belgian investigator de Meyer in 1909 proposed the name insuline, as did British researcher Schaefer in 1916.
Also Check: Is Metformin Bad For Your Heart
Insulin Resistance And Changes In The Cardiac Metabolism
The thickest layer of the heart wall is the myocardium, composed of cardiac muscle cells, thus, the knowledge provided by skeletal muscle cell physiology helps explain the cardiac metabolic function . The mammalian heart must contract incessantly which means the energy requirement for an optimal function is immense and this is an interesting phenomenon because there is no ATP reserve in heart muscle. Instead, energy is stored in cardiac muscle cells in three forms:
The first is Phosphocreatine , which can rapidly donate its high-energy phosphates to produce ATP from ADP . The energy available from PCr is relatively modest, used only during very rapid bursts of exercise .
The second is glycogen, which forms the endogenous form of energy in the cell. The muscles storage capacity for glycogen is limited. However, its advantage is that it consumes much less oxygen compared to fatty acids and is readily available for use as fuel in muscle .
The third form is triglycerides and FFA. Their oxidation is less efficient compared to glycogen, though it has greater energy input.
Depending on their availability or energy requirement , the cardiac metabolic network is highly flexible in using other substrates . The cardiomyocytes are capable of using glucose and lactate that accounts between 25 and 30% and a lesser proportion of amino acids, and ketone bodies . However, glycogen-derived glucose may contribute40% of glucose-mediated ATP production, demonstrated in rat heart .
Functional Measures Of Insulin Resistance
Another approach is to identify insulin resistant patients, based on functional markers of insulin resistance. McLoughlin et al were able to identify insulin resistant individuals from an overweight-obese cohort by looking at plasma triglyceride concentration, ratio of triglyceride to high-density lipoprotein cholesterol concentrations and insulin concentration. Using cut points of 1.47 mmol/L for triglyceride, 1.8 mmol/L for the triglyceride-high-density lipoprotein cholesterol ratio and 109 pmol/L for insulin, they achieved comparable sensitivity and specificity to the Adult Treatment Panel III to diagnose the metabolic syndrome.
Read Also: What Happens In Type 1 Diabetes
How Are Carbohydrates Protein And Fats Metabolism Interrelated
This shows the metabolic interrelationships between carbohydrates, protein and fats metabolism. Save Interrelationship Between Carbohydrate Protein Fat For Later Glycogen, a polymer of glucose, is an energy storage molecule in animals. When there is adequate ATP present, excess glucose is shunted into glycogen for storage. Glycogen is made
Relationships Between Glucose Levels And Insulin Secretion During A Glucose Challenge Test
Abstract The relationship between glucose and insulin levels was examined in a prospective study of 153 pregnant patients without diabetes who underwent a standard 50 gm glucose challenge test. One hundred eighteen women had normal screening results and 35 had abnormal screening values but a normal oral glucose tolerance test. Abnormal responders had greater insulin levels , and a higher insulin/glucose index . Patients with glucose levels less than 100 mg/dl had significantly lower insulin/glucose indices. Overall, obese patients had significantly greater glucose and insulin measurements than did nonobese women, but there was no difference within normal and abnormal groups. Glucose levels accounted for 52% of the insulin output and 29% of the insulin/glucose index variance. Neither age, parity, nor obesity contributed significantly to insulin levels in the multiple regression model. Therefore the accepted threshold for glucose screening reflects abnormal insulin output and this aberration may be indicative of the primary defect in gestational diabetes.Continue reading > >
Read Also: Can A Diabetic Have Cheese
The Basics Of High Blood Sugar
Diabetes is a problem with your body that causes blood sugar levels to rise higher than normal. This is also called hyperglycemia.
When you eat, your body breaks food down into sugar and sends it into the blood. Insulin then helps move the sugar from the blood into your cells. When sugar enters your cells, it is either used as fuel for energy right away or stored for later use. In a person with diabetes, there is a problem with insulin. But, not everyone with diabetes has the same problem.
There are different types of diabetestype 1, type 2 and gestational diabetes. If you have diabetestype 1, type 2 or gestationalyour body either doesn’t make enough insulin, can’t use the insulin well, or both.
Learn more about blood sugar Learn more about insulin
Ideal Blood Sugar Levels
A range of factors, including insulin resistance, diabetes, and an unbalanced diet, can cause blood sugar levels to spike or plummet.
The standard measurement units for blood sugar levels are milligrams per deciliter . Ideal blood sugar ranges are as follows:
| Timing |
Insulin and glucagon do not take immediate effect, particularly in people whose blood sugar levels are extremely high or low.
Also Check: Macaroni And Cheese Diabetes
Is Glucose Used To Convert Atp To Adp
However, cellular respiration is a little more difficult than just turning glucose energy into ATP. The bonds between the phosphate groups of the ATP molecule store energy. Energy is released when ATP is broken down into ADP and inorganic phosphate. Glucose is taken up by cells through proteins called transporters. Inside cells, glucose can be converted into pyruvate using enzymes and then into ATP via oxidative phosphorylation.
Therefore, glucose is used to produce ATP through an enzyme-controlled process called oxidative phosphorylation. This occurs in the mitochondria of cells. The production of energy molecules from food molecules is referred to as carbohydrate metabolism.
Glucose is also important for many other functions such as building blocks for tissues, maintaining proper blood sugar levels, and providing energy to the brain. Thus, it is not surprising that many organisms need efficient ways to use glucose as a source of energy. In fact, this is one reason why plants have evolved to produce so much glucose – to allow them to be successful even if parts of them are eaten by animals. Animals then use these nutrients within their bodies.
Some organisms can switch back and forth between glucose and oxygen as they breathe out carbon dioxide and take in oxygen. These are called aerobic organisms. Other organisms prefer to use only oxygen during exercise or other high-energy processes.

